Mark Patrick, Mouser Electronics & Jeff Schnabel, CUI Inc.
 Any electronics upon which lives depend is naturally heavily regulated and the healthcare industry is no exception. In this rapidly growing and changing sector there are a myriad of standards and guidelines that must be complied with before a product ever reaches the market. IEC60601 is the core component in the healthcare regulatory framework, encompassing many standards – all of which relate to electrical or electronic equipment intended for use in healthcare environments.
Any electronics upon which lives depend is naturally heavily regulated and the healthcare industry is no exception. In this rapidly growing and changing sector there are a myriad of standards and guidelines that must be complied with before a product ever reaches the market. IEC60601 is the core component in the healthcare regulatory framework, encompassing many standards – all of which relate to electrical or electronic equipment intended for use in healthcare environments.
The first edition of IEC60601 was published around 40 years ago and during that time has evolved to meet the changing needs of the industry, safeguarding both the medical professionals that operate the equipment and the patients to which it connects.
Currently published in its 3rd edition, the 4th edition is maturing and will become a mandatory requirement early in 2019. More than just an incremental update, this latest edition is already having a significant impact on companies designing and manufacturing medical electronics, given that the timescales for development, testing and attaining compliance certification take a significant amount of time.
One of the reasons for updating to the 4th edition is to reflect recent trends in the healthcare sector. Increasingly, health-monitoring devices are being used in the home or even as portable or wearable devices. This brings greater convenience to the patient as well as reducing pressure on often-strained medical resources, but also means greater challenges for designers. For example, many medical facilities have ‘clean’ power available for sensitive instruments, but this is not available in a home environment. Also, the fact that the devices are being used by the public (or their carers) means that interfaces have to be simplified. Finally, with the far greater prevalence of wireless technology and devices these days, electromagnetic compatibility (EMC) is an increasingly important topic, and one that is addressed in the 4th edition of IEC60601. Not only must the devices not interfere with wireless devices using technologies such as Wi-Fi and Bluetooth, the devices must not be affected by these sources of electromagnetic interference (EMI).
The core of the suite of standards is IEC60601-1, covering ‘general requirements for basic safety and essential performance’. Specific performance demands and specific types of device are covered in more depth in ‘collateral’ and ‘particular’ standards known as IEC60601-1-x and IEC60601-2-x. The 3rd edition introduced concepts such as ‘means of operator protection’ (MOOP) and ‘means of patient protection’ (MOPP) and these remain central to the 4th edition, albeit with some extensions. One completely new area for the 4th edition is the introduction of risk analysis as a requirement for medical device design.
The 4th edition extends the 40 years of work included in the 3rd edition, further enhancing its suitability for the modern environment and technologies. Among the major additions are the need to risk assess designs and also the whole area of EMC that not only addresses the myriad of wireless devices in the home and office environment, but also acknowledges that many modern medical devices communicate wirelessly with these very devices to transmit data to medical professionals for analysis.
Another area that has changed is that of equipment categories where categories such as ‘life supporting’ have now been superseded by a set of ‘intended use environments’ that are defined in collateral standard IEC60601-1-2. The definitions include:-
Professional Healthcare Facilities: Defined as areas where professional medical staff are such as hospitals, clinics, GP surgeries, dentists etc.
Home Healthcare: Defined in collateral standard IEC60601-1-11 as places where patients live. In these areas, EMC and power supply variations are challenges addressed by the standard, as is the use of the equipment by non-professional users (i.e. the patient or their carer).
Special Environments: This could be an industrial site where significant EMI may be present, for example on power lines due to heave equipment such as motors or welding. It would also apply to certain areas of hospitals where powerful equipment is being used, for example MRI or X-ray suites.
As the changes in the EMC environment due to the move of medical devices into home environments was one of the primary drivers behind the migration from the 3rd edition to the 4th edition, it should come as no surprise that significantly more emphasis is being put on EMC compliance testing. Both the testing procedures and the acceptance thresholds have been amended to suit the new needs of the market. In some cases, the changes have been significant – a comparison of test levels are set out in Figure 1 below.

Additionally, the new risk assessment procedures impact EMC as well, requiring designers to consider what type and level of disturbance can reasonably be foreseen during operation of the medical device.
The scope of IEC60601 only includes medical devices and, as such, power supplies fall outside the scope as they are not inherently medical devices. However, for designers to obtain IEC60601 approval of their devices, the overall device including any power supply must be compliant, so the EMC requirements are of concern to any supplier that wishes to sell power supplies into medical applications.
However, mains powered power supplies are likely to come under the scrutiny of IEC60601 as the voltages and / or currents present could be hazardous to patients or operators, unless sufficient isolation and separation is present in the power supply. Each MOOP/MOPP classification has a specifically defined requirement for the isolation voltage, creepage distance and insulation require to meed IEC60601. These details are shown in Figure 2.
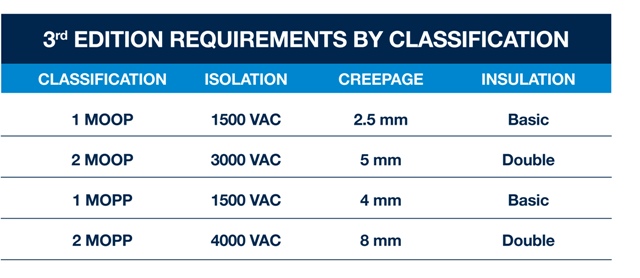
Many medical devices make direct contact with patients as part of their normal mode of operation, including blood pressure monitors, ultrasound scanners etc. These types of device must meet the 2 MOOP and 1 MOPP specifications in IEC60601. For devices such as dialysis machines and defibrillators, using power supplies that are qualified to IEC60601 is a mandatory requirement. These types of device / equipment require an additional isolation barrier between the power supply and any part of the equipment that is in direct patient contact. Designers often achieve this by using an isolation transformer, or a 2 MOPP compliant DC/DC converter with double insulation and 8mm creepage.
Sometimes, establishing which MOOP/MOPP criteria needs to be adhered to is not straightforward. While it may seem less expensive to define and implement a 1 MOPP power supply it may, in fact, be a lower cost route to implement a 2 MOPP solution thereby avoiding a redesign in the latter stages of the project, which is always an expensive proposition.
As a result of the changes coming in the 4th edition, power supply designs will change significantly. To ensure compliance, and to ensure that the isolation, leakage current and EMC performance will be acceptable, it is good practice to specify a power supply that explicitly complies with IEC60601-1 3.1 safety standards and 4th edition EMC requirements.
Given that medical safety standards are complex in nature and constantly evolving, many companies choose to outsource the power supply itself. By sourcing IEC 60601-1 4th edition compliant supplies from CUI Inc. via Mouser, designers can be assured that the power supply will be compliant and the design process will be significantly less stressful – and much shorter.






