
(Credit:http://www.powerbanksindia.com)
By Dr. S. S. Verma, Department of Physics, S.L.I.E.T., Longowal, Distt.-Sangrur (Punjab)-148106
A lithium-ion battery (abbreviated as LIB) is a type of rechargeable battery in which lithium ions move from the negative electrode to the positive electrode during discharge and back when charging. Handy, helpful lithium-ion battery was pioneered at Oxford University in the 1970s by chemist John Goodenough and his colleagues. Their research was published in 1980 and turned into a commercial technology by Sony, who produced the first lithium ion battery in the early 1990s. Since then, LiIon batteries are common in all kinds of devices. Usually, LIBs are significantly lighter than other
kinds of rechargeable batteries of similar size. Presently, the growing use of Li-Ion batteries include but are not limited to smart phones, tablets and more commonly notebooks, radio controlled hobby vehicles, portable vaporizers, UAV and drones. Some cleverly formatted Li-Ion batteries have also been used in wearable electronic devices. Li-Ion batteries are often compared to Li-Po (lithium polymer) batteries. However, Li-Po batteries aren’t commercially available due to their poor room-temperature performance. There have also been some incidents of combustion/explosion in Li-Ion batteries in high-drain applications. The best battery choice really depends on the particular application. Li-Ion has better low-temperature discharge performance, with operating ranges of -200C to 600C. Some can suffer from aging, however, even when not in use. Although care must be exercised in sourcing the batteries, Li-Ion is better suited to very high-drain devices.
Inside a lithium-ion battery
Unlike the disposable lithium primary battery, a LIB uses intercalated lithium compound instead of metallic lithium as its electrode. As with most batteries Li-ion batteries also have an outer case made of metal. The use of metal is particularly important here because the battery is pressurized. This metal case has some kind of pressure-sensitive vent hole. If the battery ever gets so hot that it risks exploding from over-pressure, this vent will release the extra pressure. The battery will probably be useless afterwards, so this is something to avoid. The vent is strictly there as a safety measure, so is the Positive Temperature Coefficient (PTC) switch, a device that is supposed to keep the battery from overheating. This metal case holds a long spiral comprising three thin sheets pressed together: positive electrode, negative electrode and a separator. Inside the case these sheets are submerged in an organic solvent that acts as the electrolyte. Ether is one common solvent. The separator is a very thin sheet of micro-perforated plastic. As the name implies, it separates the positive and negative electrodes while allowing ions to pass through. The positive electrode is made of Lithium cobalt oxide, or LiCoO2. The negative electrode is made of carbon. When the battery charges, ions of lithium move through the electrolyte from the positive electrode to the negative electrode and attach to the carbon. During discharge, the lithium ions move back to the LiCoO2 from the carbon. The movement of these lithium ions happens at a fairly high voltage, so each cell produces 3.7 volts. This is much higher than the 1.5 volts typical of a normal AA alkaline cell and helps make lithium-ion batteries more compact in small devices like cell phones. Lithium-ion battery in all shapes and sizes would have the following:
- The lithium-ion cells, either cylindrical batteries that look almost identical to AA cells, or they can be prismatic, which means they are square or rectangular.
- One or more temperature sensors to monitor the battery temperature
- A voltage converter and regulator circuit to maintain safe levels of voltage and current
- A shielded notebook connectorthat lets power and information flow in and out of the battery
- A voltage tap, which monitors the energy capacity of individual cells in the battery
- A battery charge state monitor, which is a small computer that handles the whole charging process to make sure the batteries charge as quickly and fully as possible. If the battery pack gets too hot during charging or use, the computer will shut down the flow of power to try to cool things down. In case of extremely hot environment this computer may prevent from powering up until things cool off. If the cells ever become completely discharged, the battery pack will shut down because the cells are ruined. It may also keep track of the number of charge/discharge cycles and send out information on how much charge is left in the battery.
Working of a lithium-ion battery
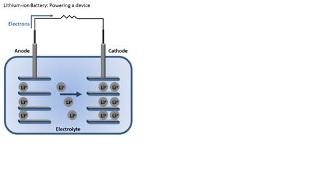 Batteries store and release energy by moving electrons from one end of the battery to the other. Then we can use the energy from those moving electrons to do work for us. These two battery “ends” are known as electrodes. One is called the anode and the other is called the cathode. Generally, the anode is made from carbon and the cathode from a chemical compound known as a metal oxide (cobalt oxide, for example). The final battery ingredient is known as the electrolyte, and it sits in between the two electrodes. In the case of lithium-ion batteries, the electrolyte is a salt solution that contains lithium ions—hence the name. When we place the battery in a device, the positively charged lithium ions are attracted to and move towards the cathode. Once it is bombarded with these ions, the cathode becomes more positively charged than the anode, and this attracts negatively charged electrons. As the electrons start moving toward the cathode, we force them to go through our device and use the energy of the electrons “flowing” toward the cathode to generate power. You can think of this kind of like a water wheel, except instead of water flowing, electrons are flowing. Lithium-ion batteries are great because they are rechargeable. When the battery is connected to a charger, the lithium ions move in the opposite direction as before. As they move from the cathode to the anode, the battery is restored for another use.
Batteries store and release energy by moving electrons from one end of the battery to the other. Then we can use the energy from those moving electrons to do work for us. These two battery “ends” are known as electrodes. One is called the anode and the other is called the cathode. Generally, the anode is made from carbon and the cathode from a chemical compound known as a metal oxide (cobalt oxide, for example). The final battery ingredient is known as the electrolyte, and it sits in between the two electrodes. In the case of lithium-ion batteries, the electrolyte is a salt solution that contains lithium ions—hence the name. When we place the battery in a device, the positively charged lithium ions are attracted to and move towards the cathode. Once it is bombarded with these ions, the cathode becomes more positively charged than the anode, and this attracts negatively charged electrons. As the electrons start moving toward the cathode, we force them to go through our device and use the energy of the electrons “flowing” toward the cathode to generate power. You can think of this kind of like a water wheel, except instead of water flowing, electrons are flowing. Lithium-ion batteries are great because they are rechargeable. When the battery is connected to a charger, the lithium ions move in the opposite direction as before. As they move from the cathode to the anode, the battery is restored for another use.
Growth & development of Li-Ion batteries
Lithium battery technology has taken many years to develop. It offers distinct advantages over other older rechargeable battery technologies such as Nickel cadmium and Nickel Metal Hydride. The idea for lithium ion battery technology was first proposed in the 1970s by M Whittingham who used titanium sulphide and lithium metal as the electrodes for his cell. Work was undertaken at the University of Pennsylvania to enable a graphite electrode to be used with lithium ions in the electrode. This was a major advance, although it was not taken up immediately by other advances in lithium ion technology. However other techniques associated with charging needed to be solved before a viable cell could be made. In 1979 J Goodenough demonstrated a rechargeable lithium ion cell using Lithium Cobalt Oxide for the positive electrode and lithium as the negative one. The next stages in producing a workable production cell were to be able to enable recharging action with lithium in graphite which was achieved by in 1979. It then took until 1985 before a rechargeable lithium ion cell was developed that could be manufactured in large scale production quantities. Akira Yoshino used carbonaceous material which would accept lithium ions as one electrode, and lithium cobalt oxide, LiCoO2 as the other. The use of Lithium Cobalt Oxide was important because it is stable in air unlike lithium itself, and this made this cell structure more stable chemically and far less dangerous. Lithium-ion batteries are common in home and portable electronics. LIBs are also growing in popularity for military, battery electric vehicle and aerospace applications. Chemistry, performance, cost and safety characteristics vary across LIB types. Handheld electronics mostly use LIBs based on lithium cobalt oxide (LiCoO2), which offers high energy density, but presents safety risks, especially when damaged. Lithium iron phosphate (LiFePO4), lithium ion manganese oxide battery (LiMn2O4, Li2MnO3, or LMO) and lithium nickel manganese cobalt oxide (LiNiMnCoO2 or NMC) offer lower energy density, but longer lives and less likelihood of explosion. Such batteries are widely used for electric tools, medical equipment, and other roles. Lithium nickel cobalt aluminum oxide (LiNiCoAlO2 or NCA) and lithium titanate (Li4Ti5O12 or LTO) are specialty designs aimed at particular niche roles. The newer lithium–sulfur batteries promise the highest performance-to-weight ratio. Lithium-ion batteries can pose unique safety hazards since they contain a flammable electrolyte and may be kept pressurized.
A brief history of research and technology advances reaching to the present state of art of lithium ion battery is as:
| LITHIUM ION BATTERY TECHNOLOGIES SUMMARY | |||
| NAME | CONSTITUENTS | MAJOR CHARACTERISTICS | APPLICATIONS |
| Lithium Cobalt | LiCoO2 | High capacity | Cellphones, laptops, cameras |
| Lithium Manganese Oxide | LiMn2O4 | Safety, but lower capacity | Power tools, medical, hobbyist |
| Lithium Iron Phosphate | LiFePO4 | Safety, but lower capacity | Power tools, medical, hobbyist |
| Lithium Nickel Manganese Cobalt Oxide | LiNiMnCoO2 | Safety, but lower capacity | Power tools, medical, hobbyist |
| Lithium Nickel Cobalt Aluminium Oxide | LiNiCoAlO2 | Electric vehicles and grid storage | |
| Lithium Titanate | Li4Ti5O12 | Electric vehicles and grid storage | |
In order to improve the efficiency and decrease the charge time of lithium-ion batteries, many companies and researchers are using nanotechnology to make better battery materials. A lot of research is focused on using nanotechnology to make better electrodes. Using nanomaterials in the electrodes increases their surface area, which provides more places for the lithium ions to make contact. This makes the battery more efficient and also makes it recharge faster. These changes should make electronic devices that use lithium ion batteries lighter and also allow them to go a longer time before recharging. Lithium-ion batteries using nanophosphate for the cathode material take advantage of the increased surface area provided by nanomaterials. This allows the battery to go through thousands of charges, an estimated two to three times more than other lithium ion batteries, without changes in performance. In addition to the longer life, the nanophosphate batteries are much lighter than other lithium-ion batteries and charged fast.
New designs
Lithium-air batteries — believed to be able to hold up to five times more energy than the lithium-ion batteries that power our phones, laptops and electric vehicles — have been tantalizing to battery researchers for years but several obstacles have plagued this development. The batteries would work by combining lithium present in the anode with oxygen from the air to produce lithium peroxide on the cathode during the discharge phase. The lithium peroxide would be broken back down into its lithium and oxygen components during the charge phase. Unfortunately, experimental designs of such lithium-air batteries have been unable to operate in a true natural-air environment due to the oxidation of the lithium anode and production of undesirable byproducts on the cathode that result from lithium ions combining with carbon dioxide and water vapor in the air. These byproducts gum up the cathode, which eventually becomes completely coated and unable to function. These experimental batteries have relied on tanks of pure oxygen — which limits their practicality and poses serious safety risks due to the flammability of oxygen. The research team overcame these challenges by using a unique combination of anode, cathode and electrolyte — the three main components of any battery — to prevent anode oxidation and buildup of battery-killing byproducts on the cathode and allow the battery to operate in a natural-air environment. They coated the lithium anode with a thin layer of lithium carbonate that selectively allows lithium ions from the anode to enter the electrolyte while preventing unwanted compounds from reaching the anode. In a lithium-air battery, the cathode is simply where the air enters the battery. In experimental designs of lithium-air batteries, oxygen, together with all the other gases that make up air, enters the electrolyte through a carbon-based spongy lattice structure. Researchers coated the lattice structure with a molybdenum disulfate catalyst and used a unique hybrid electrolyte made of ionic liquid and dimethyl sulfoxide, a common component of battery electrolytes, that helped facilitate lithium-oxygen reactions, minimize lithium reactions with other elements in the air and boost efficiency of the battery. Researchers have designed a new lithium-air battery that works in a natural-air environment and still functioned after a record-breaking 750 charge/discharge cycles.
Advantages of lithium-ion batteries
Lithium-ion batteries (LIBs) are incredibly popular these days. Today, lithium-ion is one of the most successful and safe battery chemistries available. Two billion cells are produced every year. We can find them in laptops, PDAs, cell phones and iPods. They’re some of the most energetic rechargeable batteries available. Lithium-ion batteries are popular because they have a number of important advantages over competing technologies:
Lighter: much lighter than other types of rechargeable batteries of the same size. The electrodes of a lithium-ion battery are made of lightweight lithium and carbon.
High-energy density: Lithium is also a highly reactive element, meaning that a lot of energy can be stored in its atomic bonds. This translates into a very high energy density for lithium-ion batteries. A typical lithium-ion battery can store 150 watt-hours of electricity in 1 kilogram of battery. For a comparison, a NiMH (nickel-metal hydride) battery pack can store perhaps 100 watt-hours per kilogram, although 60 to 70 watt-hours might be more typical. A lead-acid battery can store only 25 watt-hours per kilogram. Using lead-acid technology, it takes 6 kilograms to store the same amount of energy that a 1 kilogram lithium-ion battery can handle.
Long charge holding: They hold their charge for long and lose only about 5 percent of its charge per month, compared to a 20 percent loss per month for NiMH batteries. Lithium-ion batteries can handle hundreds of charge/discharge cycles.
Self-discharge: One issue with batteries and cells is that they lose their charge over time. This self-discharge can be a major issue. One advantage of lithium ion cells is that their rate of self-discharge is much lower than that of other rechargeable cells such as Ni-Cad and NiMH forms.
Memory effect: They have no memory effect, which means that we do not have to completely discharge them before recharging, as with some other battery chemistries.
Rechargeable: Lithium ion battery is rechargeable makes it more desirable and sustainable.
Variety of types available: There are several types of lithium ion cell available. This advantage of lithium ion batteries can mean that the right technology can be used for the particular application needed. Some forms of lithium ion battery provide a high current density and are ideal for consumer mobile electronic equipment. Others are able to provide much higher current levels and are ideal for power tools and electric vehicles.
Problems with lithium-ion batteries
That is not to say that lithium-ion batteries are flawless. They have a few problems as well:
- They start degrading as soon as they leave the factory. They will only last two or three years from the date of manufacture whether we use them or not.
- They are extremely sensitive to high temperatures. Heat causes lithium-ion battery packs to degrade much faster than they normally would.
- If we completely discharge a lithium-ion battery, it is ruined.
- A lithium-ion battery pack must have an on-board computer to manage the battery. This makes them even more expensive than they already are.
- Lithium-ion batteries have the possibility to burst into flames occasionally. Though this is not very common — just two or three battery packs per million have a problem.
- Protection required: lithium ion cells and batteries are not as robust as some other rechargeable technologies. They require protection from being over charged and discharged too far. In addition to this, they need to have the current maintained within safe limits. Accordingly one lithium ion battery disadvantage is that they require protection circuitry incorporated to ensure they are kept within their safe operating limits. Fortunately with modern integrated circuit technology, this can be relatively easily incorporated into the battery or within the equipment if the battery is not interchangeable.
- Ageing: One of the major lithium ion battery disadvantages for consumer electronics is that lithium ion batteries suffer from ageing. Not only is this time or calendar dependent, but it is also dependent upon the number of charge discharge cycles that the battery has undergone. When a typical consumer lithium cobalt oxide, LCO battery or cell needs to be stored it should be partially charged – around 40% to 50% and kept in a cool storage area. Storage under these conditions will help increase the life.
- Transportation: Another disadvantage of lithium ion batteries is that there can be certain restrictions placed on their transportation, especially by air. Although the batteries that could be taken in aircraft carry-on luggage are unlikely to be affected, care should be taken not to carry any more lithium ion batteries than are needed. Any carried separately must be protected against short circuits by protective covers, etc.
- Cost: A major lithium ion battery disadvantage is their cost. Typically they are around 40% more costly to manufacture than Nickel cadmium cells. This is a major factor when considering their use in mass produced consumer items where any additional costs are a major issue.
- Immature technology: Lithium ion battery technology is a developing area. This can be a disadvantage in terms of the fact that the technology does not remain constant. However as new lithium ion technologies are being developed all the time, it can also be an advantage as better solutions are coming available.
Guidelines to use Li-ion batteries
Lithium-ion batteries are expensive, so to make it last longer, here are some things to keep in mind:
- Avoid heat, which degrades the batteries
- Li-ion batteries should be bought only when needed, because the aging process begins as soon as the battery is manufactured. If a Li-ion battery is not to be used for an extended period of time it should ideally be brought to a charge level of between about 40% and 60% of full charge.
- Li-ion batteries should be kept cool. By keeping them cool, possibly in a refrigerator, the ageing process becomes slower. As a result, Li-ion batteries should not be kept in cars on sunny days as the temperatures rise significantly
- Li-ion batteries should never be depleted to below their minimum voltage, 2.4 V to 3.0 V per cell
- Li-ion batteries should not be exposed to very low temperatures – most lithium-ion battery electrolytes freeze at approximately 40°C. This may preclude them from some applications where equipment needs powering in extremes of temperature
- Lithium ion chemistry prefers partial dischargeto deep discharge, so it’s best to avoid taking the battery all the way down to zero. Since lithium-ion chemistry does not have a “memory”, we do not harm the battery pack with a partial discharge. If the voltage of a lithium-ion cell drops below a certain level, it’s ruined
- Lithium-ion batteries should be charged before the battery is completely discharged
- Lithium-ion batteries should not be frequently fully discharged and recharged (“deep-cycled”) like Ni-Cd batteries. However this may be needed occasionally to recalibrate any associated “fuel gauge” circuitry used to monitor the state of charge, control charging, etc.
Acknowledgement: The use of information retrieved through various references/sources of internet in this article is highly acknowledged.






